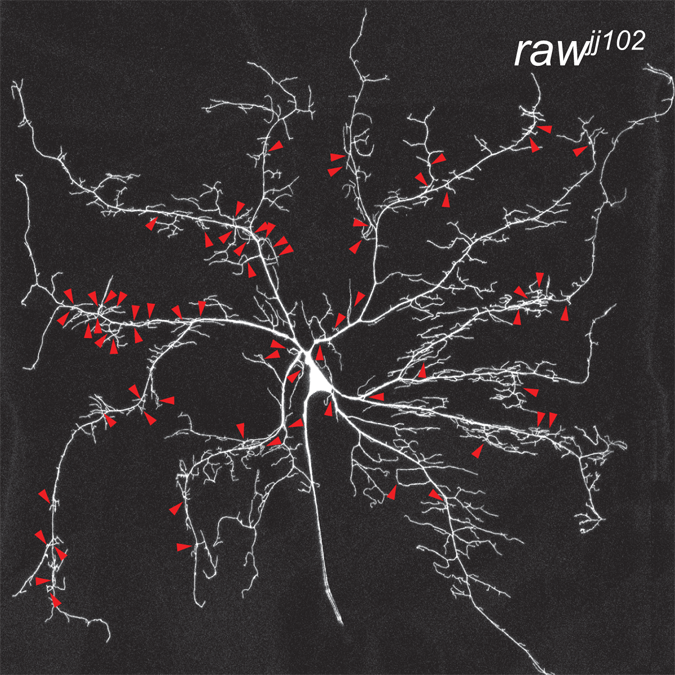
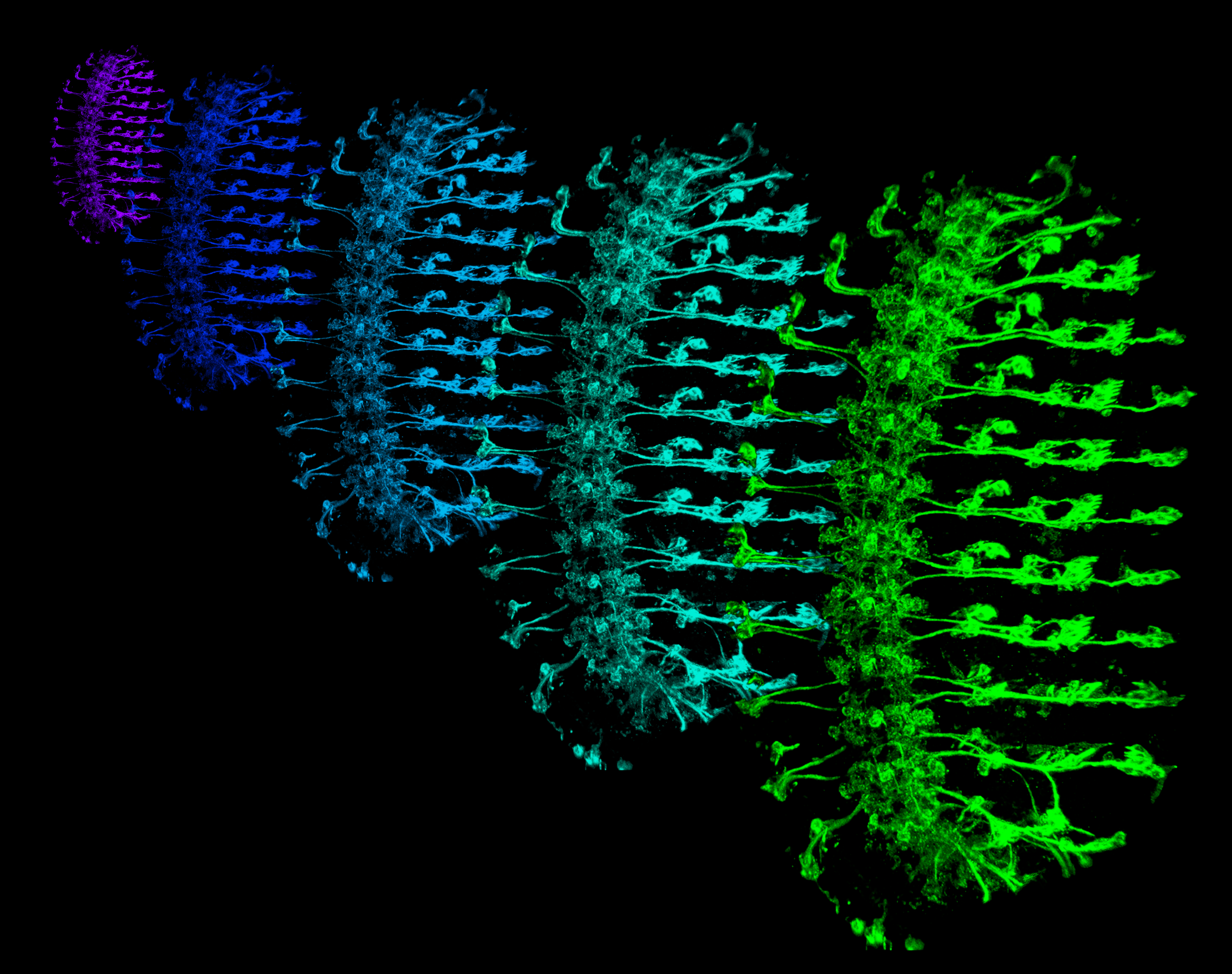
Somatosensory neurons (SSNs) that detect and transduce mechanical, thermal, and chemical stimuli densely innervate an animal’s skin. However, although epidermal cells provide the first point of contact for sensory stimuli, our understanding of roles that epidermal cells play in SSN function, particularly nociception, remains limited.
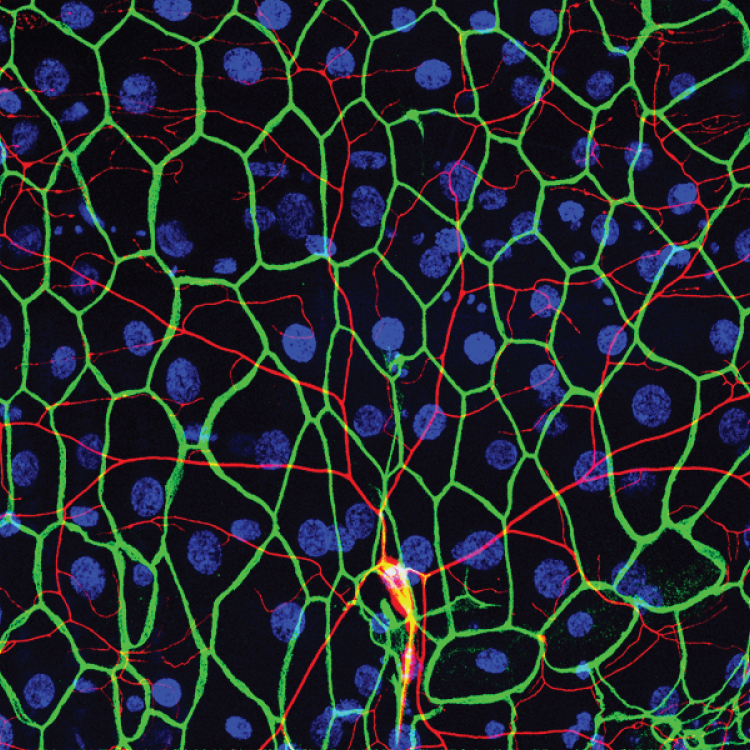
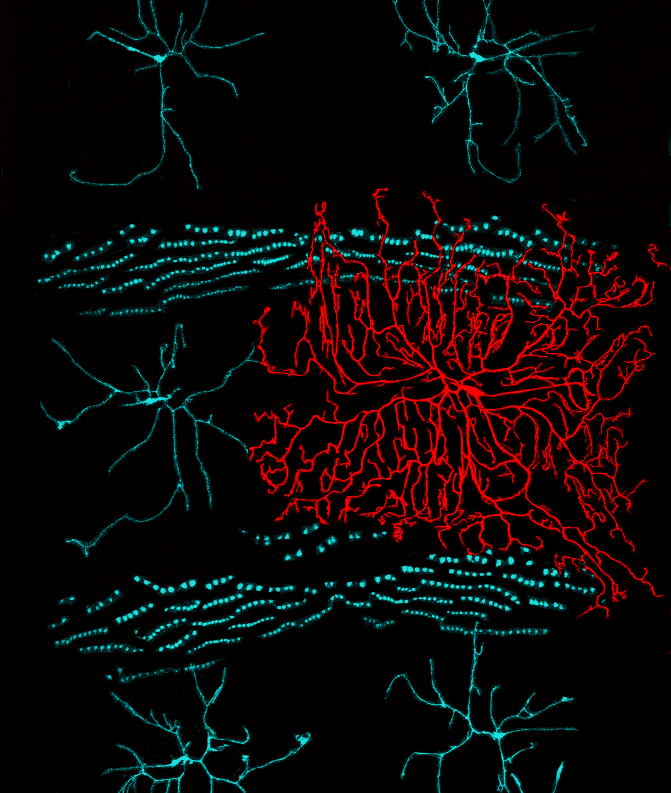
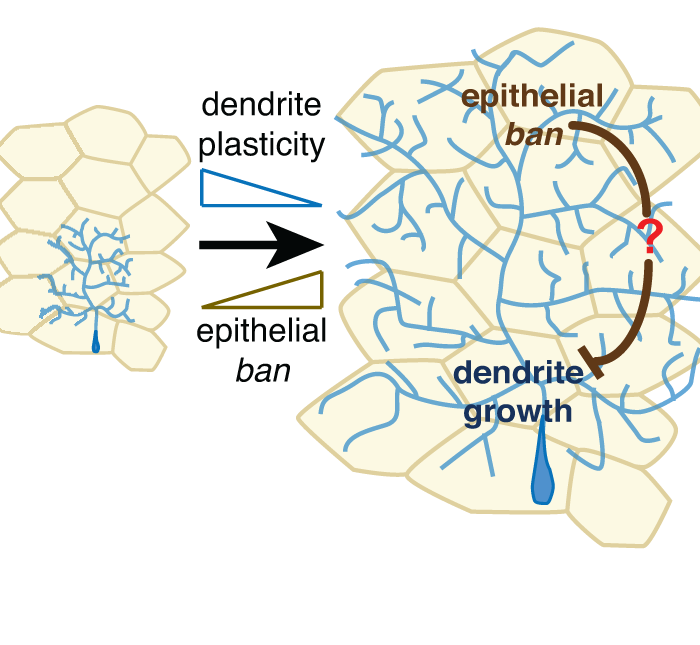
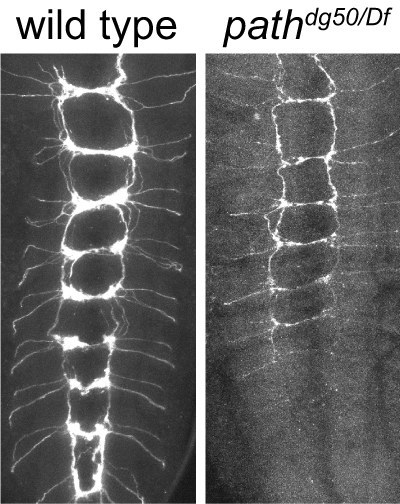
Recent studies demonstrate that epidermal cells work in concert with SSNs to transduce noxious and innocuous physical stimuli. For example, epidermal Merkel cells are mechanosensory cells that signal to sensory neurons to mediate touch transduction. Similarly, keratinocytes are directly activated by noxious thermal and mechanical stimuli and release molecules that modulate nociceptor functions. Furthermore, epidermal cells in invertebrates and vertebrates ensheathe nociceptors in mesaxon-like structures, and these sheaths may serve as sites of epidermis-nociceptor signaling. Indeed, epidermal ensheathment is required for normal responses to noxious mechanical stimuli in Drosophila.
Using Drosophila larvae as our experimental system we are examining sensory functions of resident skin cells and their contribution to animal behavior.
Mechanosensory function of epidermal cells
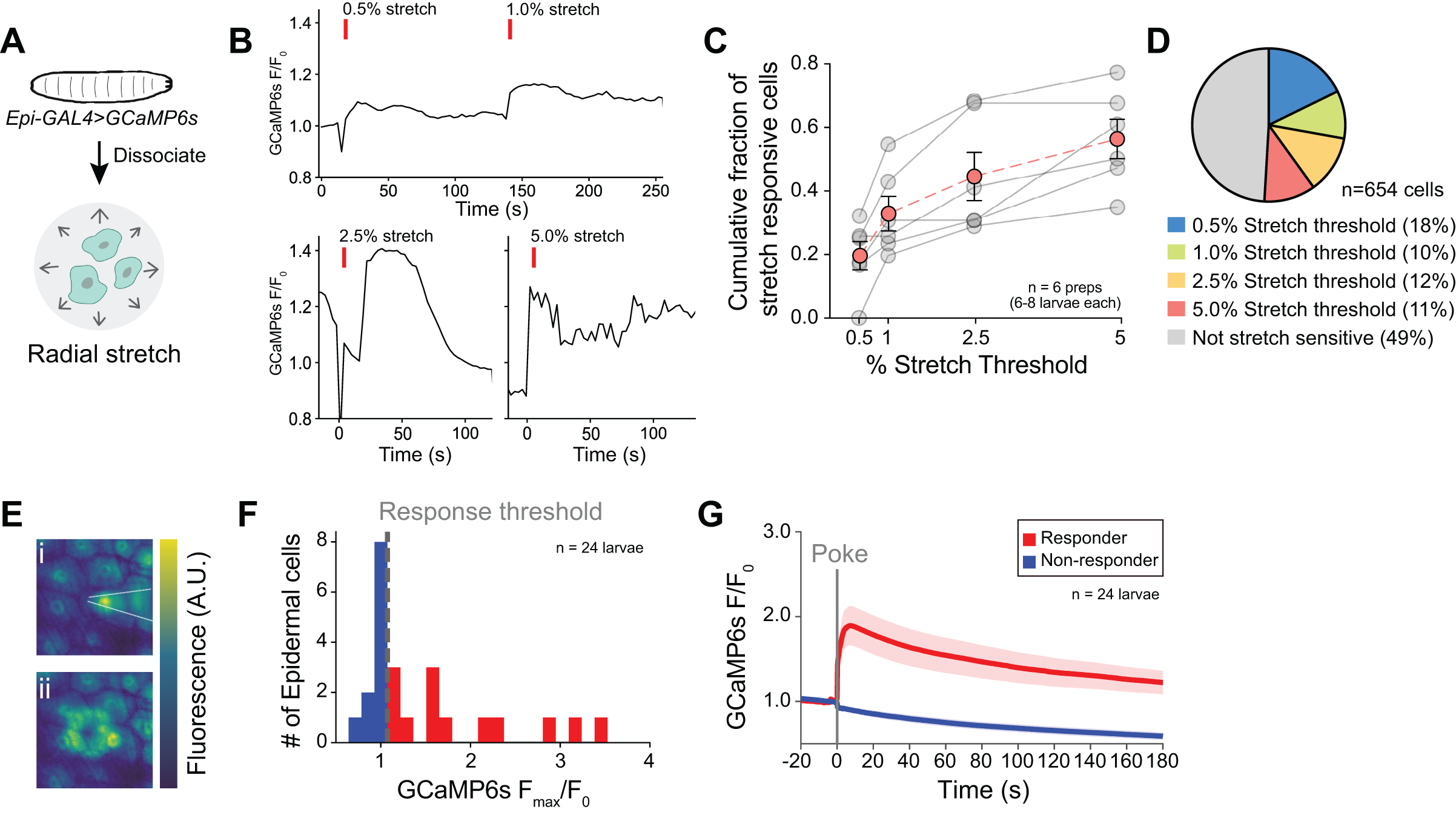
We found that optogenetic or thermogenetic stimulation of epidermal cells evokes activity in a variety of SSNs neurons and triggers complex behavioral responses, suggesting that epidermal cells can modulate activity of SSNs in Drosophila. Prior studies have shown that vertebrate epidermal cells directly respond to mechanical stimuli, therefore we assessed whether Drosophila epidermal cells are intrinsically mechanosensitive. Indeed, we found that epidermal cells responded to a variety to mechanosensory inputs including radial stretch, hypoosmotic challenge, and laminar flow.
Thermosensory responses of epidermal cells